Scientists have achieved a groundbreaking first—capturing a 3D view of killer T cells in action as they destroy cancer cells. This new perspective reveals an astonishing level of precision and organization, where T cells form a tiny contact zone that delivers a lethal blow without harming healthy neighbors. The discovery highlights a hidden molecular choreography that could revolutionize our understanding of immune responses and inspire next-generation cancer therapies. Below, we answer key questions about this remarkable process.
How do killer T cells locate and identify their targets?
Killer T cells, also known as cytotoxic T lymphocytes, patrol the body like elite security guards. They rely on special receptors on their surface that scan for antigens—fragments of proteins displayed on the surface of other cells. Cancer cells often present abnormal antigens due to mutations. When a T cell's receptor locks onto a matching antigen on a cancer cell, it initiates a tight binding. This recognition step is highly specific: the T cell will only engage if the antigen fits perfectly, ensuring that healthy cells without such markers are left untouched. Once identified, the T cell prepares to deploy its lethal machinery.
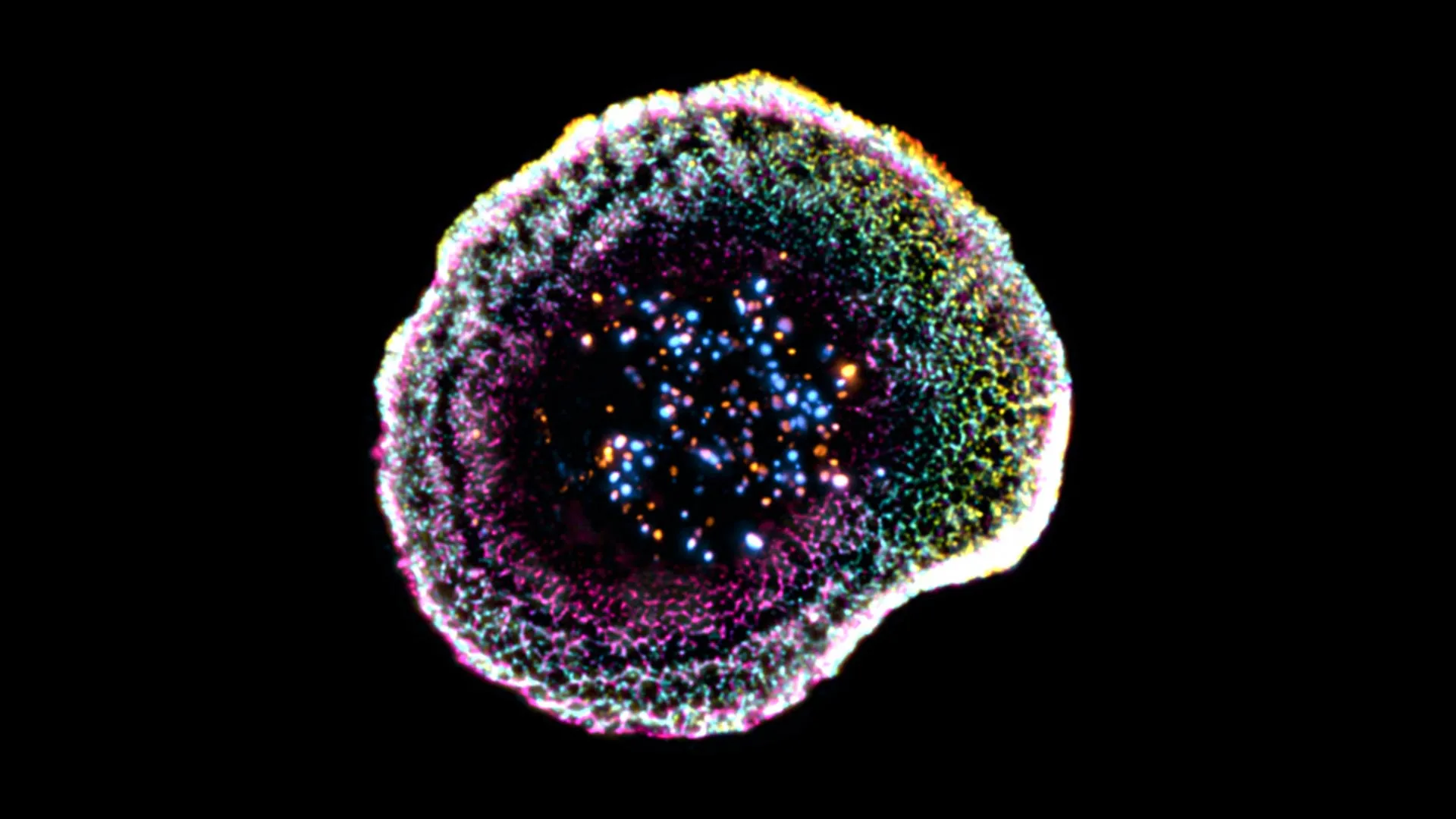
What is the 'contact zone' and why is it so critical?
The contact zone is a highly organized interface that forms between the killer T cell and its target. Think of it as a carefully constructed molecular docking station. At this spot, the T cell concentrates key proteins—such as perforin and granzymes—into a small area. Perforin punches holes in the target cell's membrane, while granzymes enter those holes to trigger cell death. Without this precise organization, the toxic molecules could leak out and damage neighboring cells, causing collateral harm. The contact zone ensures that destruction is confined to the intended target, exemplifying the immune system's remarkable precision.
How did scientists achieve the first-ever 3D view of this process?
Until now, observing this microscopic ballet was limited to 2D snapshots or blurry images. The breakthrough came from a technique called cryo-electron tomography (cryo-ET). Researchers rapidly froze T cells and cancer cells together at the moment of attack, preserving natural structures. Then, they used electron beams to record thousands of tilt-series images, which were computationally reconstructed into a detailed 3D model. This allowed them to zoom in on the contact zone at near-atomic resolution, revealing the spatial arrangement of proteins and membranes that had never been seen before. The study, published in Nature, marks a leap in visualizing immune cell interactions.
What new details did the 3D view reveal about the molecular choreography?
The 3D images unveiled a surprising level of order. Instead of a random jumble, the contact zone showed distinct layers: a central area packed with signaling receptors, surrounded by a ring of adhesion molecules that anchor the cells together, and an outer ring where pore-forming proteins cluster. This arrangement ensures that perforin is released precisely at the center, directly over the target cell's membrane. The study also revealed that T cells form small protrusions that probe the target surface, almost like fingers checking for weak spots. This choreography optimizes the delivery of deadly payloads while minimizing the risk of harming bystander cells.
Why is this discovery important for cancer treatment?
Understanding the exact geometry of T cell attacks provides a blueprint for improving immunotherapies. Many current treatments, such as CAR-T cell therapy, engineer a patient's T cells to better recognize cancer. However, they don't always form an efficient contact zone. With this 3D knowledge, scientists can tweak the molecular adhesive forces or the clustering of killing machinery to make T cells more effective. Additionally, the insights could help design drugs that stabilize the contact zone, reducing the chance of treatment failure. Ultimately, this research opens a new window into how we can harness the body's own defenses with greater precision, potentially reducing side effects and boosting survival rates.
Could this research lead to new immunotherapies?
Absolutely. The detailed 3D view acts as a roadmap for innovation. For instance, researchers might engineer T cells to express adhesion molecules that strengthen the contact zone, or to deliver perforin more efficiently. Another possibility is to create bispecific antibodies that link T cells to cancer cells, mimicking the natural docking process. Additionally, the findings could inform the development of checkpoint inhibitors that remove brakes on T cell activity—if we know how the contact zone forms, we can better predict why some tumors resist therapy. The study also inspires new imaging techniques to monitor T cell performance in patients. In short, this molecular choreography is a goldmine for designing smarter, more targeted cancer immunotherapies.